AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Helium electric configuration12/16/2023 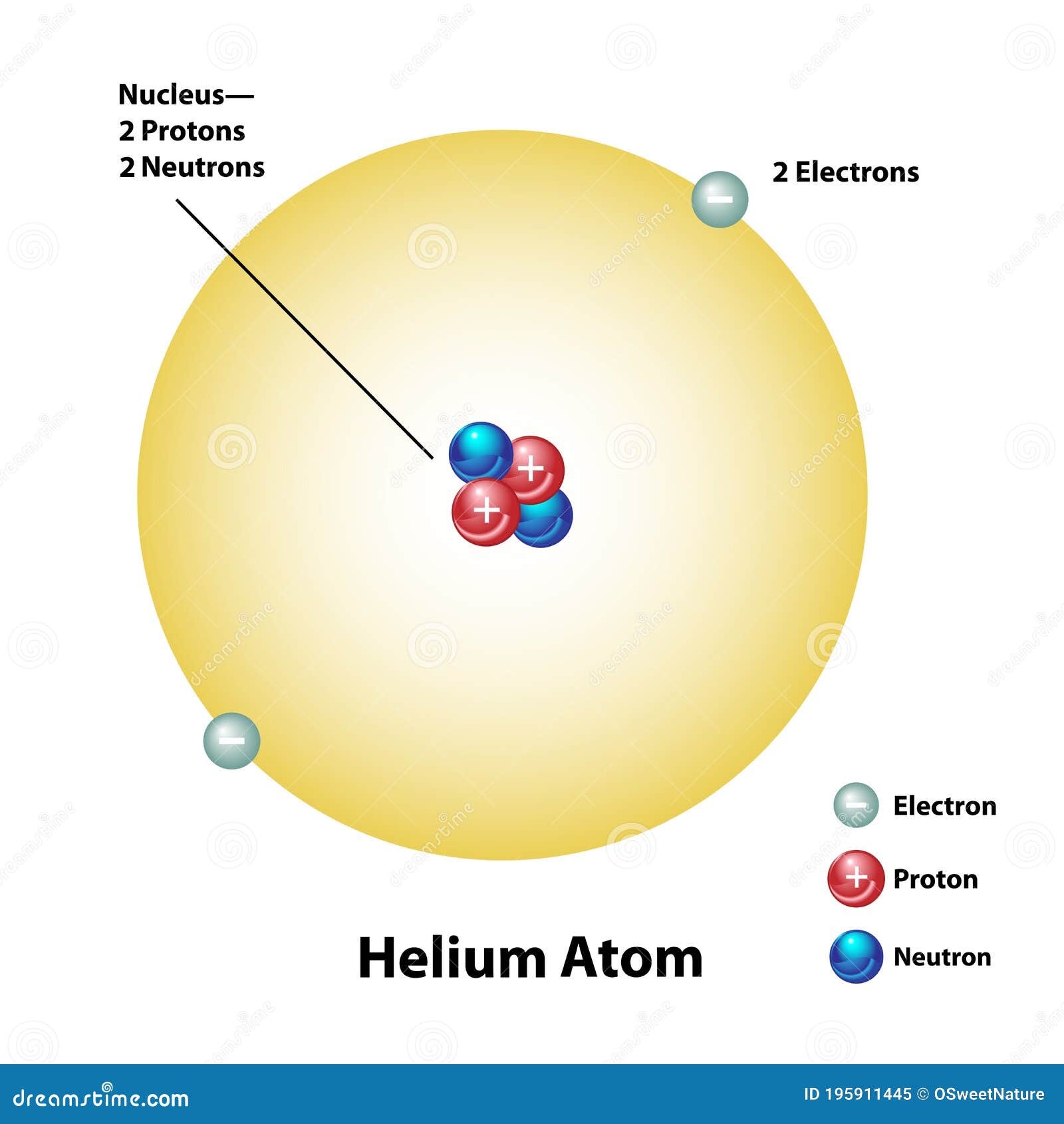
To find the electron configuration of an element, start at hydrogen and trace across each period until your target element is reached. Writing Electron Configurations – Examples Groups or blocks of the periodic table share the same sublevel, and are divided as seen in the following diagram. Sublevels are indicated by letters s, p, d, and f. 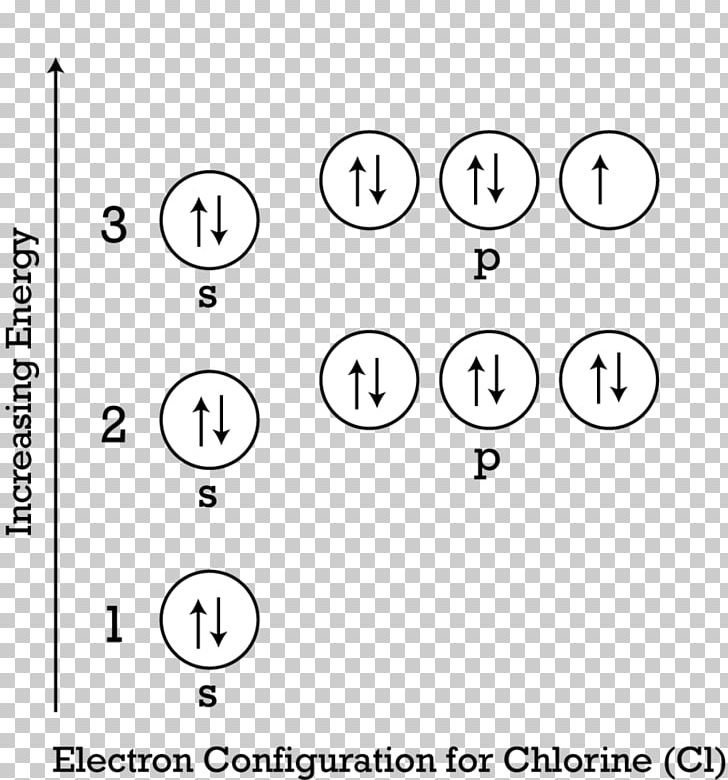
Each successive integer generally represents a higher energy level than the last. The principle energy level is indicated by an integer (1, 2, 3, …7) that corresponds with the periods on the periodic table. The periodic table is a helpful tool in writing these configurations. Lithium, containing three electrons, has two electrons occupying an s orbital at the first energy level, and one electron occupying an s orbital at the second energy level. The electron configuration for Lithium is: 1s 2 2s 1 Systems with a greater number of electrons will occupy a greater amount of energy levels. In this case, there are two electrons in an s orbital with the principle energy level of one. The first integer, 1, gives us the principle energy level, the letter s represents the type of orbital (sublevel), and the superscript 2 gives us the electron occupancy. Here is the electron configuration for Helium: 1 s 2 Writing Electron ConfigurationsĮlectron configurations have a standard notation that tells you the principle energy levels and sublevels that electrons occupy. Electron configurations are represented by standard written notation, or by using orbital diagrams. Systems with a greater number of electrons will occupy a greater amount of energy levels, meaning that they also will utilize higher energy levels. Electrons occupy orbitals that have characteristic levels of energy. The electron configuration is a description of where electrons are in a molecule or atom. If you enjoy this tutorial, be sure to check out our others linked below! Related Articles You will learn Aufbau’s principle, Hund’s rule and the Pauli exclusion principle. In this tutorial, you will learn how to find and write the electron configuration and orbital diagram for various elements using the periodic table.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
